Abstract
Research Article
Novel extractive visible spectrophotometric method for determination of antihypertensive drug irbesartan with sulfonephthalein acid dyes in tablets
Safwan Ashour* and Roula Bayram
Published: 22 June, 2022 | Volume 6 - Issue 1 | Pages: 006-012
Irbesartan (IRB) is one of the drugs used for the treatment of hypertension. The present work develops and validates two methods for the evaluation of irbesartan in bulk and tablets. Sulfonephthalein acid dyes, bromophenol blue (BPB), and bromocresol purple (BCP) were used to produce stable yellow ion-association complexes with the basic drug IRB in dry chloroform. The colored products are quantified spectrophotometrically at their corresponding λmax. The relation between the absorbed signal and the drug concentration was linear up to 45.0 μg mL-1 (n = 6,
r ≥ 0.9998). LOD reaches 40 ng mL-1. The composition of the ion associates was found 1:1 by Job’s and mole ratio methods. Application of the suggested methods to dosage forms is presented with percentage recoveries ranging from 99.33% to 101.67%. The results of the analysis were validated statistically and compared with the official method. No interference was observed from common pharmaceutical adjuvants.
Read Full Article HTML DOI: 10.29328/journal.apps.1001029 Cite this Article Read Full Article PDF
Keywords:
Extractive spectrophotometry; Irbesartan; Bromophenol blue (BPB); Bromocresol purple (BCP); Pharmaceuticals
References
- British Pharmacopœia, Her Majesty Stationery Officer, London, UK, 2013.
- Cazaubon C, Gougat J, Bousquet F, Guiraudou P, Gayraud R, Lacour C, Roccon A, Galindo G, Barthelemy G, Gautret B, Bernhart C, Perreaut P, Breliere JC, LeFur G, Nisto D, Pharmacological characterization of SR47436, a new nonpeptide AT1 subtype angiotensin II receptor antagonist. J Pharmacol Exp Ther 265(2), 1993, 826–834.ISSN 1521-0103
- United States Pharmacopoeial Convention Inc. Rockville, 2011.
- Rahman N, Siddiqui MR, Azmi SN. Quantitative analysis of irbesartan in commercial dosage forms by kinetic spectrophotometry. Chem Pharm Bull (Tokyo). 2006 May;54(5):626-31. doi: 10.1248/cpb.54.626. PMID: 16651756.
- Ashour S, Bayram R, Selective and validated kinetic spectrophotometric method for the determination of irbesartan in pure and pharmaceutical formulations, Annales Pharmaceutiques Françaises, 77(2), 2019, 101–111. https://doi.org/10.1016/j.pharma.2018.09.002
- El-Shaboury SR, Hussein SA, Mohamed NA, El-Sutohy MM. Spectrofluorimetric method for determination of some angiotensin II receptor antagonists. J Pharm Anal. 2012 Feb;2(1):12-18. doi: 10.1016/j.jpha.2011.10.005. Epub 2011 Nov 10. PMID: 29403715; PMCID: PMC5760827.
- El-Bagary RI, Hashem HM, Waleed A, Ebeid WA, Spectrofluorometric, spectrophotometric and LC determination of irbesartan. Journal of Chemical and Pharmaceutical Research, 3(4), 2011, 722–733. https://www.jocpr.com/articles/septrofluorometric-spectrophotometric-and-lc-determination-of-irbesartan.pdf. ISSN No: 0975-7384.
- Ganesh K, Balraj C, Elango KP. Spectroscopic and spectrofluorimetric studies on the interaction of irbesartan with 2,3-dichloro-5,6-dicyano-1,4-benzoquinone and iodine. Spectrochim Acta A Mol Biomol Spectrosc. 2011 Sep;79(5):1621-9. doi: 10.1016/j.saa.2011.05.022. PMID: 21684193.
- Kulsum S, Padmalatha M, Kumar E, Sruthi M, Vidyasagar G, Spectrophotometric methods for the determination of irbesartan in pure and pharmaceutical dosage forms. Research Journal of Pharmacy and Technology, 2011, 4(10), 1567–1569.
- Nissankararao S, Anil Kumar A, Bhimavarapu RD, Krishna Prasanna V, Estimation of irbesartan in bulk and dosage forms by new simple UV spectrophotometry using hydrotropic technique. Pharmaceutica Analytica Acta. 2013, 4(8): 8–10.https://doi.org/10.4172/2153-2435.1000265
- Li JM, Wu Q, Wang LL, Assay of irbesartan tablets by HPLC. Chinese Journal of Pharmaceutical Analysis, 2001, 21(4), 249–250.
- Goswami N. A validated stability-indicating liquid chromatographic method for determination of process related impurities and degradation behavior of Irbesartan in solid oral dosage. J Adv Pharm Technol Res. 2014 Jan;5(1):33-40. doi: 10.4103/2231-4040.126985. PMID: 24695518; PMCID: PMC3960792.
- Ganesan M, Nanjundan S, Gomathi M, Muralidharan S, Method development and validation of Irbesartan using LCMS/MS: Application to pharmacokinetic studies. Journal of Chemical and Pharmaceutical Research, 2(4), 2010, 740–746. ISSN No: 0975-7384
- Praveen Kumar M, Sreeramulu J, Development and validation of a stability-indicating RP-HPLC method for assay of irbesartan in pure and pharmaceutical dosage form. International Journal of Pharmaceutical Sciences Review and Research, 6(10), 2011, 94–99. ISSN 0976 – 044X
- Sane RT, Francis M, Pawar S, Determination of irbesartan in pharmaceutical dosage forms by HPTLC. Indian Drugs, 2002, 39(1), 32–35.
- Patil SH, Janjale MV. Novel and validated titrimetric method for determination of selected angiotensin-II-receptor antagonists in pharmaceutical preparations and its comparison with UV spectrophotometric determination. J Pharm Anal. 2012 Dec;2(6):470-477. doi: 10.1016/j.jpha.2012.03.009. Epub 2012 Apr 9. PMID: 29403786; PMCID: PMC5760927.
- Gupta VK, Jain R, Agarwal S, Mishra R, Dwivedi A. Electrochemical determination of antihypertensive drug irbesartan in pharmaceuticals. Anal Biochem. 2011 Mar 15;410(2):266-71. doi: 10.1016/j.ab.2010.11.024. Epub 2010 Nov 19. PMID: 21094117.
- Vetuschi C, Giannandrea A, Carlucci G, Mazzeo P. Determination of hydrochlorothiazide and irbesartan in pharmaceuticals by fourth-order UV derivative spectrophotometry. Farmaco. 2005 Aug;60(8):665-70. doi: 10.1016/j.farmac.2005.04.013. PMID: 15963513.
- Joseph Charles J, Brault S, Boyer C, Langlois MH, Cabrero L, Dubost JP, Simultaneous determination of irbesartan and hydrochlorothiazide in tablets by derivative spectrophotometry, Analytical Letters, 36(11), 2003, 2485–2495. https://doi.org/10.1081/AL-120024337
- Erk N. Three new spectrophotometric methods applied to the simultaneous determination of hydrochlorothiazide and irbesartan. Pharmazie. 2003 Aug;58(8):543-8. PMID: 12967029.
- Albero I, Ródenas V, García S, Sánchez-Pedreño C. Determination of irbesartan in the presence of hydrochlorothiazide by derivative spectrophotometry. J Pharm Biomed Anal. 2002 Jun 20;29(1-2):299-305. doi: 10.1016/s0731-7085(02)00073-0. PMID: 12062690.
- Sivasubramanian L, Lakshmi KS, Spectrophotometric multicomponent analysis of irbesartan, hydrochlorothiazide and ramipril in pharmaceutical formulations by chemometric techniques. Journal of Analytical & Pharmaceutical Research 2(3), 2016, 1–10. DOI: 10.15406/japlr.2016.02.00019
- Üstündağ Ö, Dinç E, Simultaneous quantitative resolution and validation of a binary mixture in a pharmaceutical dosage form by using a spectrophotometric signal processing technique. J. Fac. Pharm. Ankara, 44(3), 2020, 506–516. Doi: 10.33483/jfpau.799897
- Abdel Monem Hegazy M, Fayez YM. Mean centering of ratio spectra and concentration augmented classical least squares in a comparative approach for quantitation of spectrally overlapped bands of antihypertensives in formulations. Spectrochim Acta A Mol Biomol Spectrosc. 2015 Apr 5;140:210-5. doi: 10.1016/j.saa.2014.12.103. Epub 2015 Jan 5. PMID: 25594214.
- Fayez YM. Simultaneous determination of some anti-hypertensive drugs in their binary mixture by novel spectrophotometric methods. Spectrochim Acta A Mol Biomol Spectrosc. 2014 Nov 11;132:446-51. doi: 10.1016/j.saa.2014.04.102. Epub 2014 Apr 30. PMID: 24887506.
- Lakshmi S, Lakshmi KS, H-point standard addition method for simultaneous spectrophotometric determination of irbesartan, hydrochlorothiazide and telmisartan in tablets. International J. Res. Pharm. Chem. 4(2), 2014, 373– ISSN: 2231-2781
- Hillaert S, Van den Bossche W. Simultaneous determination of hydrochlorothiazide and several angiotensin-II-receptor antagonists by capillary electrophoresis. J Pharm Biomed Anal. 2003 Feb 26;31(2):329-39. doi: 10.1016/s0731-7085(02)00643-x. PMID: 12609672.
- Hillaert S, Van den Bossche W. Optimization and validation of a capillary zone electrophoretic method for the analysis of several angiotensin-II-receptor antagonists. J Chromatogr A. 2002 Dec 6;979(1-2):323-33. doi: 10.1016/s0021-9673(02)01247-5. PMID: 12498264.
- Alanazi AM, Abdelhameed AS, Khalil NY, Khan AA, Darwish IA, HPLC method with monolithic column for simultaneous determination of irbesartan and hydrochlorothiazide in tablets. Acta Pharmaceutica, 2014, 64(2), 187–198. https://doi.org/10.2478/acph-2014-0014
- Rane VP, Patil KR, Sangshetti JN, Yeole RD, Shinde DB. Stability indicating LC method for simultaneous determination of irbesartan and hydrochlorothiazide in pharmaceutical preparations. J Chromatogr Sci. 2010 Aug;48(7):595-600. doi: 10.1093/chromsci/48.7.595. PMID: 20819286.
- Vujić Z, Mulavdić N, Smajić M, Brborić J, Stankovic P. Simultaneous analysis of irbesartan and hydrochlorothiazide: an improved HPLC method with the aid of a chemometric protocol. Molecules. 2012 Mar 16;17(3):3461-74. doi: 10.3390/molecules17033461. PMID: 22426527; PMCID: PMC6268177.
- Koyuturk S, Can NO, Atkosar Z, Arli G. A novel dilute and shoot HPLC assay method for quantification of irbesartan and hydrochlorothiazide in combination tablets and urine using second generation C18-bonded monolithic silica column with double gradient elution. J Pharm Biomed Anal. 2014 Aug;97:103-10. doi: 10.1016/j.jpba.2014.04.026. Epub 2014 May 2. PMID: 24876066.
- KurbanoĞlu S, Yarman A. Simultaneous Determination of Hydrochlorothiazide and Irbesartan from Pharmaceutical Dosage Forms with RP-HPLC. Turk J Pharm Sci. 2020 Oct;17(5):523-527. doi: 10.4274/tjps.galenos.2019.76094. Epub 2020 Oct 30. PMID: 33177933; PMCID: PMC7650730.
- Ibrahim AE, Elmansi H, Belal F. Solvent-free mixed micellar mobile phases: An advanced green chemistry approach for reversed-phase HPLC determination of some antihypertensive drugs. J Sep Sci. 2020 Aug;43(16):3224-3232. doi: 10.1002/jssc.202000429. Epub 2020 Jun 23. PMID: 32510825.
- Ali TA, Mohamed GG, Aglan AA, Heakal FE, RP-HPLC stability-indicating method for estimation of irbesartan and hydrochlorothiazide in bulk and pharmaceutical dosage form. Chinese Journal of Analytical Chemistry, 44(1), 2016, e1601–e1608. https://doi.org/10.1016/S1872-2040(16)60899-X
- Zаrеh MM, Sааd MZ, Hаssаn WS, Еlhеnnаwу MЕ, Soltan MK, Sеbаiу MM. Grаdiеnt HPLC Mеthоd fоr Simultаnеоus Dеtеrminаtiоn оf Еight Sаrtаn аnd Stаtin Drugs in Thеir Purе аnd Dоsаgе Fоrms. Pharmaceuticals (Basel). 2020 Feb 20;13(2):32. doi: 10.3390/ph13020032. PMID: 32093384; PMCID: PMC7169415.
- Alhazmi HA, Alnami AM, Arishi MAA, Alameer RK, Al Bratty M, Rehman ZU, Javed SA, Arbab IA. A Fast and Validated Reversed-Phase HPLC Method for Simultaneous Determination of Simvastatin, Atorvastatin, Telmisartan and Irbesartan in Bulk Drugs and Tablet Formulations. Sci Pharm. 2017 Dec 19;86(1):1. doi: 10.3390/scipharm86010001. PMID: 29257120; PMCID: PMC5874531.
- Abdellatef HE. Extractive-spectrophotometric determination of disopyramide and irbesartan in their pharmaceutical formulation. Spectrochim Acta A Mol Biomol Spectrosc. 2007 Apr;66(4-5):1248-54. doi: 10.1016/j.saa.2006.06.015. Epub 2006 Jun 27. PMID: 16920393.
- Ashour S, Bayram R, Sensitive extractional colorimetric analysis of fexofenadine hydrochloride and irbesartan bases through acid-dye complexation using naphthol blue black in pure form and pharmaceuticals, Modern Chemistry, 2017, 5(6), 93–http:/doi=10.11648/j.mc.20170506.12
- Ashour S, Chehna MF, Bayram R. Spectrophotometric Determination of Alfuzosin HCl in Pharmaceutical Formulations with some Sulphonephthalein Dyes. Int J Biomed Sci. 2006 Sep;2(3):273-8. PMID: 23674990; PMCID: PMC3614605.
- Ashour S, Al-Khalil R. Simple extractive colorimetric determination of levofloxacin by acid-dye complexation methods in pharmaceutical preparations. Farmaco. 2005 Sep;60(9):771-5. doi: 10.1016/j.farmac.2005.06.007. PMID: 16023643.
- Rose J, Advanced physico-chemical experiments, Pittman, London, 1964, 67.
- Martin AN, Swarbrick J, Cammarata A. Physical Pharmacy. Third Edition, Lee and Febiger, Philadelphia, 1983, 344–346.
- Miller JN, Miller JC, Statistics and Chemometrics For Analytical Chemistry, Chapman & Hall/CRC, London, UK, 5th edition, 2005.
- Ringbom A, Zeitschrift Analytische Chemie, 1939, pp. 115–332.
- Long GL, Winefordner JD. Limit of Detection. A Closer Look at the IUPAC Definition, Anal. Chem. 1983, 55(7), 712A–724A. https://doi.org/10.1021/ac00258a724
Figures:
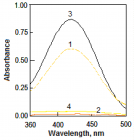
Figure 1
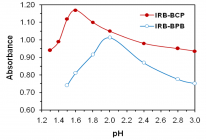
Figure 2
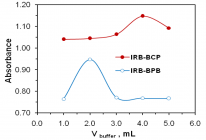
Figure 3
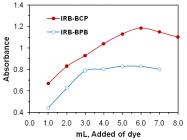
Figure 4
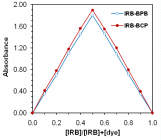
Figure 5
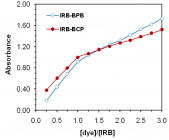
Figure 6
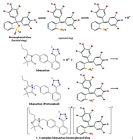
Figure 7
Similar Articles
-
Novel extractive visible spectrophotometric method for determination of antihypertensive drug irbesartan with sulfonephthalein acid dyes in tabletsSafwan Ashour*,Roula Bayram. Novel extractive visible spectrophotometric method for determination of antihypertensive drug irbesartan with sulfonephthalein acid dyes in tablets. . 2022 doi: 10.29328/journal.apps.1001029; 6: 006-012
Recently Viewed
-
A Low-cost High-throughput Targeted Sequencing for the Accurate Detection of Respiratory Tract PathogenChangyan Ju, Chengbosen Zhou, Zhezhi Deng, Jingwei Gao, Weizhao Jiang, Hanbing Zeng, Haiwei Huang, Yongxiang Duan, David X Deng*. A Low-cost High-throughput Targeted Sequencing for the Accurate Detection of Respiratory Tract Pathogen. Int J Clin Virol. 2024: doi: 10.29328/journal.ijcv.1001056; 8: 001-007
-
A Comparative Study of Metoprolol and Amlodipine on Mortality, Disability and Complication in Acute StrokeJayantee Kalita*,Dhiraj Kumar,Nagendra B Gutti,Sandeep K Gupta,Anadi Mishra,Vivek Singh. A Comparative Study of Metoprolol and Amlodipine on Mortality, Disability and Complication in Acute Stroke. J Neurosci Neurol Disord. 2025: doi: 10.29328/journal.jnnd.1001108; 9: 039-045
-
Development of qualitative GC MS method for simultaneous identification of PM-CCM a modified illicit drugs preparation and its modern-day application in drug-facilitated crimesBhagat Singh*,Satish R Nailkar,Chetansen A Bhadkambekar,Suneel Prajapati,Sukhminder Kaur. Development of qualitative GC MS method for simultaneous identification of PM-CCM a modified illicit drugs preparation and its modern-day application in drug-facilitated crimes. J Forensic Sci Res. 2023: doi: 10.29328/journal.jfsr.1001043; 7: 004-010
-
A Gateway to Metal Resistance: Bacterial Response to Heavy Metal Toxicity in the Biological EnvironmentLoai Aljerf*,Nuha AlMasri. A Gateway to Metal Resistance: Bacterial Response to Heavy Metal Toxicity in the Biological Environment. Ann Adv Chem. 2018: doi: 10.29328/journal.aac.1001012; 2: 032-044
-
Obesity in Patients with Chronic Obstructive Pulmonary Disease as a Separate Clinical PhenotypeDaria A Prokonich*, Tatiana V Saprina, Ekaterina B Bukreeva. Obesity in Patients with Chronic Obstructive Pulmonary Disease as a Separate Clinical Phenotype. J Pulmonol Respir Res. 2024: doi: 10.29328/journal.jprr.1001060; 8: 053-055
Most Viewed
-
Evaluation of Biostimulants Based on Recovered Protein Hydrolysates from Animal By-products as Plant Growth EnhancersH Pérez-Aguilar*, M Lacruz-Asaro, F Arán-Ais. Evaluation of Biostimulants Based on Recovered Protein Hydrolysates from Animal By-products as Plant Growth Enhancers. J Plant Sci Phytopathol. 2023 doi: 10.29328/journal.jpsp.1001104; 7: 042-047
-
Sinonasal Myxoma Extending into the Orbit in a 4-Year Old: A Case PresentationJulian A Purrinos*, Ramzi Younis. Sinonasal Myxoma Extending into the Orbit in a 4-Year Old: A Case Presentation. Arch Case Rep. 2024 doi: 10.29328/journal.acr.1001099; 8: 075-077
-
Feasibility study of magnetic sensing for detecting single-neuron action potentialsDenis Tonini,Kai Wu,Renata Saha,Jian-Ping Wang*. Feasibility study of magnetic sensing for detecting single-neuron action potentials. Ann Biomed Sci Eng. 2022 doi: 10.29328/journal.abse.1001018; 6: 019-029
-
Pediatric Dysgerminoma: Unveiling a Rare Ovarian TumorFaten Limaiem*, Khalil Saffar, Ahmed Halouani. Pediatric Dysgerminoma: Unveiling a Rare Ovarian Tumor. Arch Case Rep. 2024 doi: 10.29328/journal.acr.1001087; 8: 010-013
-
Physical activity can change the physiological and psychological circumstances during COVID-19 pandemic: A narrative reviewKhashayar Maroufi*. Physical activity can change the physiological and psychological circumstances during COVID-19 pandemic: A narrative review. J Sports Med Ther. 2021 doi: 10.29328/journal.jsmt.1001051; 6: 001-007

HSPI: We're glad you're here. Please click "create a new Query" if you are a new visitor to our website and need further information from us.
If you are already a member of our network and need to keep track of any developments regarding a question you have already submitted, click "take me to my Query."